Overview
Why Is A Stroke Diagnostic So Important?
Almost half of patients going to Emergency Departments with symptoms consistent with stroke, actually have another problem that looks like a stroke, but isn’t. Of the patients with an actual stroke, ~13% have a hemorrhagic stroke, which is diagnosed via CT scan. The other 87% have an ischemic stroke, which is usually not detectable by a CT scan during the acute time period, when treatment to break up the clot provides the greatest benefits. This leaves doctors with a critical diagnostic gap: how can we very quickly distinguish patients who look like they are having a stroke from those who actually are having a stroke? Time is of the essence: every second, 30,000 neurons are lost. The use of the powerful clot-busting drug (e.g., tissue Plasminogen Activator (tPA)) must be provided rapidly in the acute presenting window to be effective but treatment with this drug on those not having a stroke are exposed to the risk of developing a bleed, the added costs from tPA therapy including a required hospital stay, and delay of diagnosis and treatment of the true issue.
Acute Brain Injury IVD for Stroke
TETmedical has developed the NSE-FAST (Functional Activity Stroke Test), an In Vitro Diagnostic (IVD) for stroke to be used with standard plate readers. The test fits into standard clinical practice in the Emergency Department, with a potential high sensitivity and negative predictive value that would be highly useful with significant potential payer cost savings and reduced patient risk.
The NSE-FAST is in development and clinical studies and is not cleared or approved by the U.S. FDA for diagnostic use.
Given the speed and sensitivity attainable with our diagnostic platform, TET is focused on measuring the levels of Neuron Specific Enolase functional activity (NSE-FA) which is released rapidly into the blood by damaged neurons during a brain injury like a stroke.
TETMedical’s Revolutionary Approach
Addressing this gap is so important that scientists and doctors have been trying for decades to identify “biomarkers,” molecules in the bloodstream that will reveal the presence of dying and damaged brain cells. Because it is found in large amounts in each neuron, the protein Neuron-specific Enolase (NSE) has been well-studied as a candidate biomarker. Unfortunately, these efforts to detect NSE for this use have been relatively unsuccessful as the tests were antibody based and measure the concentration of the protein. Antibody-based detection methods have a number of problems:
- they often suffer from low signal:noise ratios reducing their sensitivity,
- their results vary depending on the antibodies used,
- antibodies cannot differentiate between NSE as a functionally active enzyme that has a <6 hour half life and when it loses activity to become a protein that lasts for may days, and
- they tend to take long-periods of time to perform.
Because of the speed and sensitivity attainable with the TET diagnostic platform, we measure newly released NSE through its activity. Unlike antibodies, TETmedical’s NSE-FAST has intrinsic signal amplification at the earliest stage. In <5 minutes from the time that plasma is added to the wells, the TET reaction reveals the amount of recently-released active NSE. A pilot pre-clinical study in a stroke center showed a clear difference between patients who had a brain injury and those presenting with stroke mimics, all using a standard hospital-based plate reader.*
NSE-FAST Does Not Change Standard of Care Stroke Workflow
Today – patients presenting at the hospital with stroke symptom as seen in the diagram below, have a blood draw for standard blood tests than are assessed clinically based on their symptoms with an often used NIH stroke scale (NIHSS) as a quantifier for symptoms. A non-contrast CT scan is then run to identify bleeding associated with a hemorrhagic stroke. If the CT is negative (about 85% of the time) the neurologist and ER doctors must then decide if it is really a stroke from a blood clot (an ischemic stroke) or just a stroke mimic from something else. Many common symptoms can be vague and unspecific to stroke. Without a biomarker to identify “real” brain injury, many patients are misdiagnosed and treated with clot busters (i.e. TPA), putting them at risk due to causing bleeds.
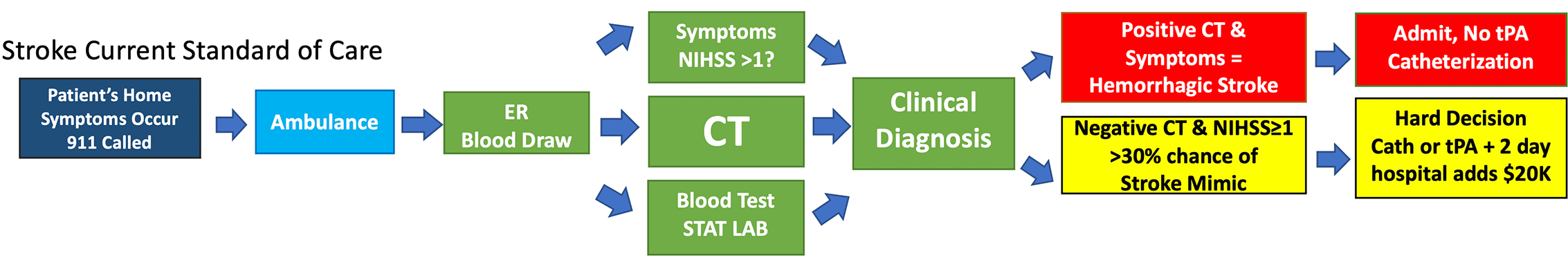
Just as the diagnosis of heart attack has been revolutionized by the diagnostic assays for the biomarker High Sensitivity Troponin (current market over half a billion dollars/year), the TET NSE-FAST could revolutionize the detection of stroke.
About Enzymes
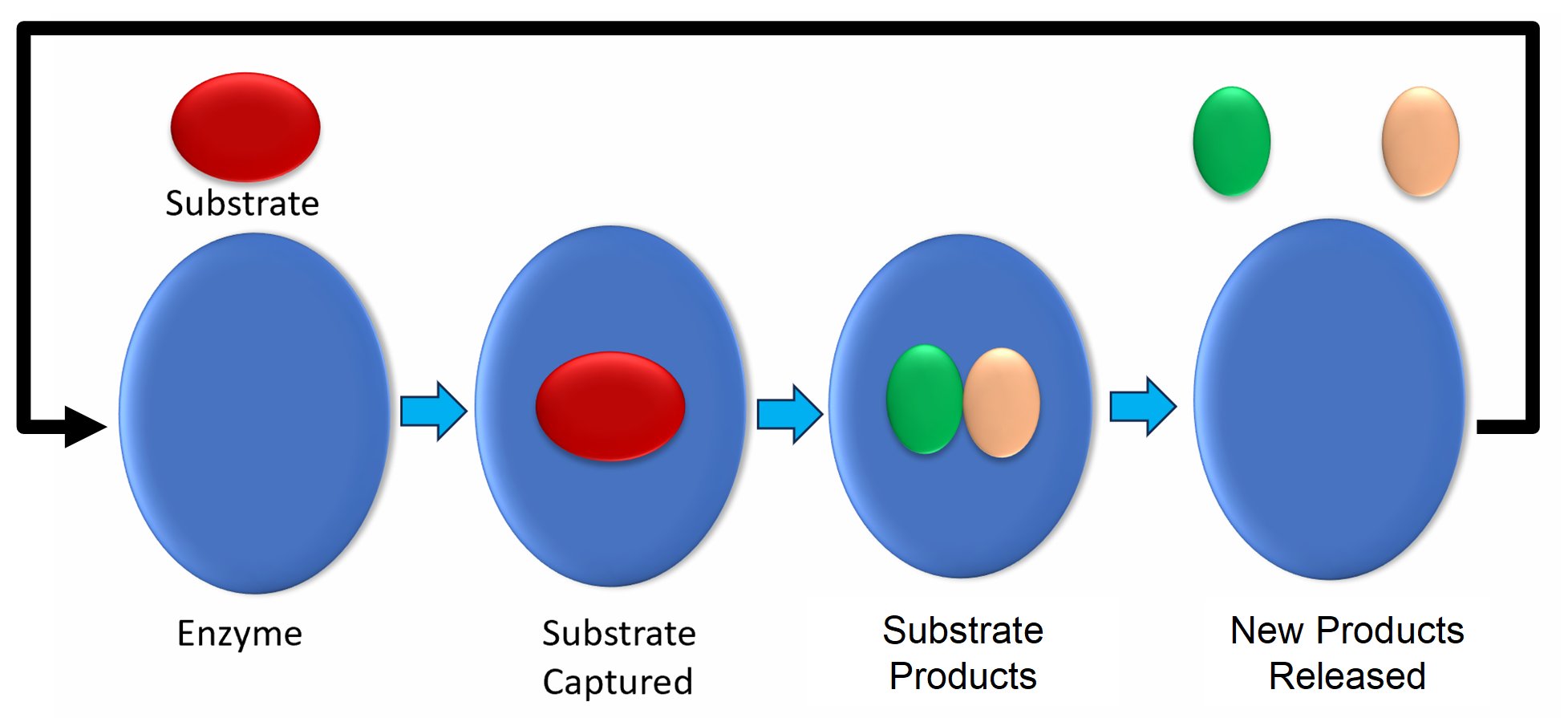
Enzymes are nature’s assembly line workers that as shown in the figure below, they attach to an organic compound or substrate, change it, release the result and do it again up to a thousand times a second.
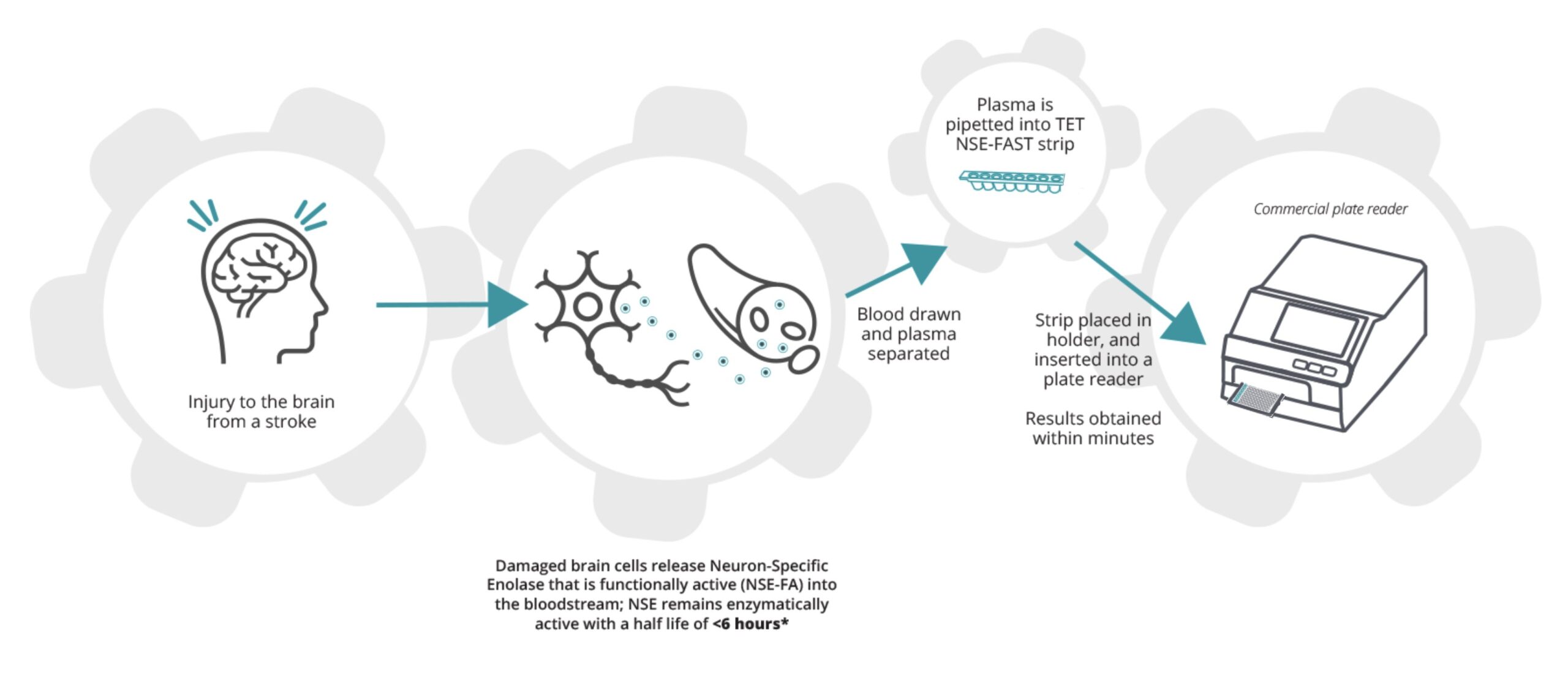
How the NSE-FAST Works
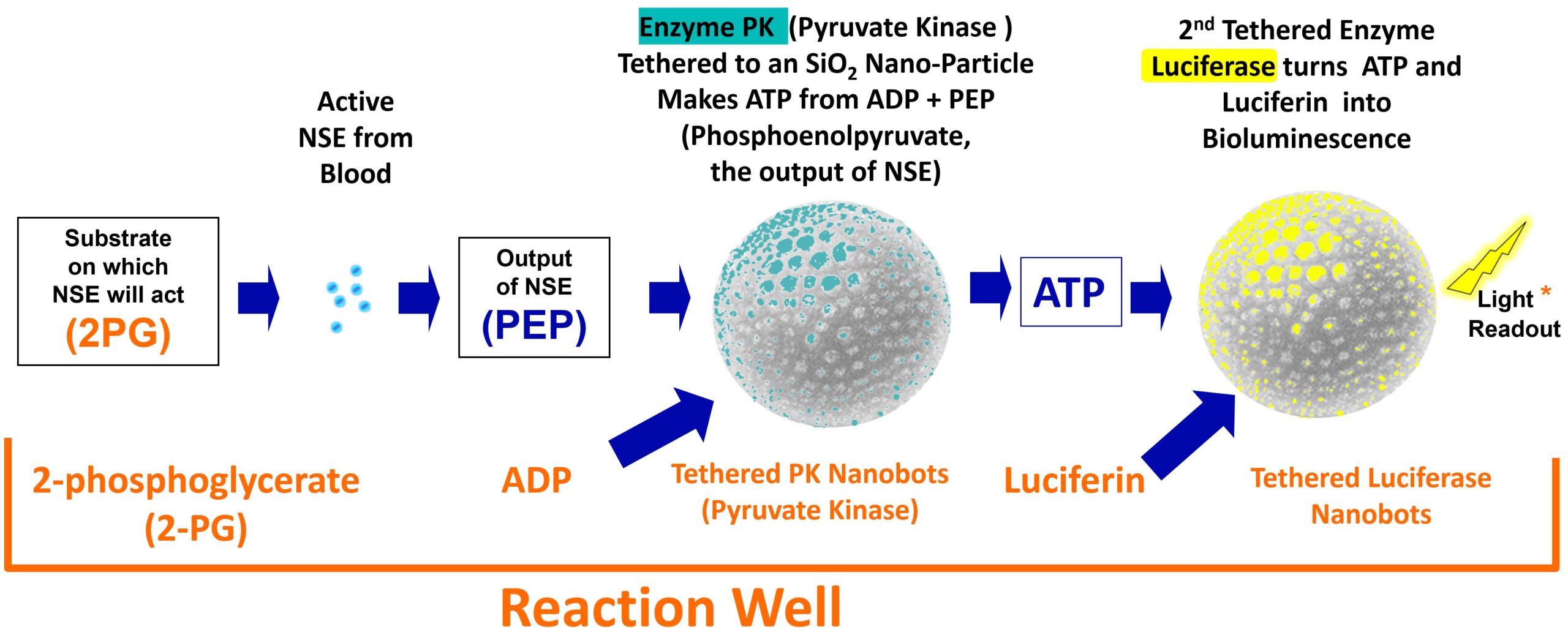
When there is injury to the brain from a stroke damaged neurons release cell contents into the bloodstream including the active enzyme NSE that makes up about 4% of the contents of a neuron. NSE remains active in the blood with a half life of <6 hours when it loses its activity but remains as a protein. Blood drawn and spun down to plasma. About 20 microliters of plasma is pipetted into each of the 8 wells in the TET NSE-FAST strip shown below. The strip that has 3 test reaction wells, 3 positive control wells and 2 negative control wells is placed in an 8 strip holder that is then inserted into a commercial plate reader that uses the luminescence from the reaction well compared to the positive and negative control wells to measure the amount of active NSE in the sample.
The NSE-FAST test reaction well contents are shown below. They include 2-PG, the substrate that the enzyme NSE takes in and changes to PEP, ADP, Luciferin and two tethered enzyme nanobots with a thousand or more enzymes tethered to a silica nanoparticle with the active side facing outward. The first nanobot has the enzyme PK that used the PEP produced by NSE to change ADP to ATP. The 2nd nanobot is Luciferase that allows fireflies to light up that takes ADP and Luciferin to emit photons.
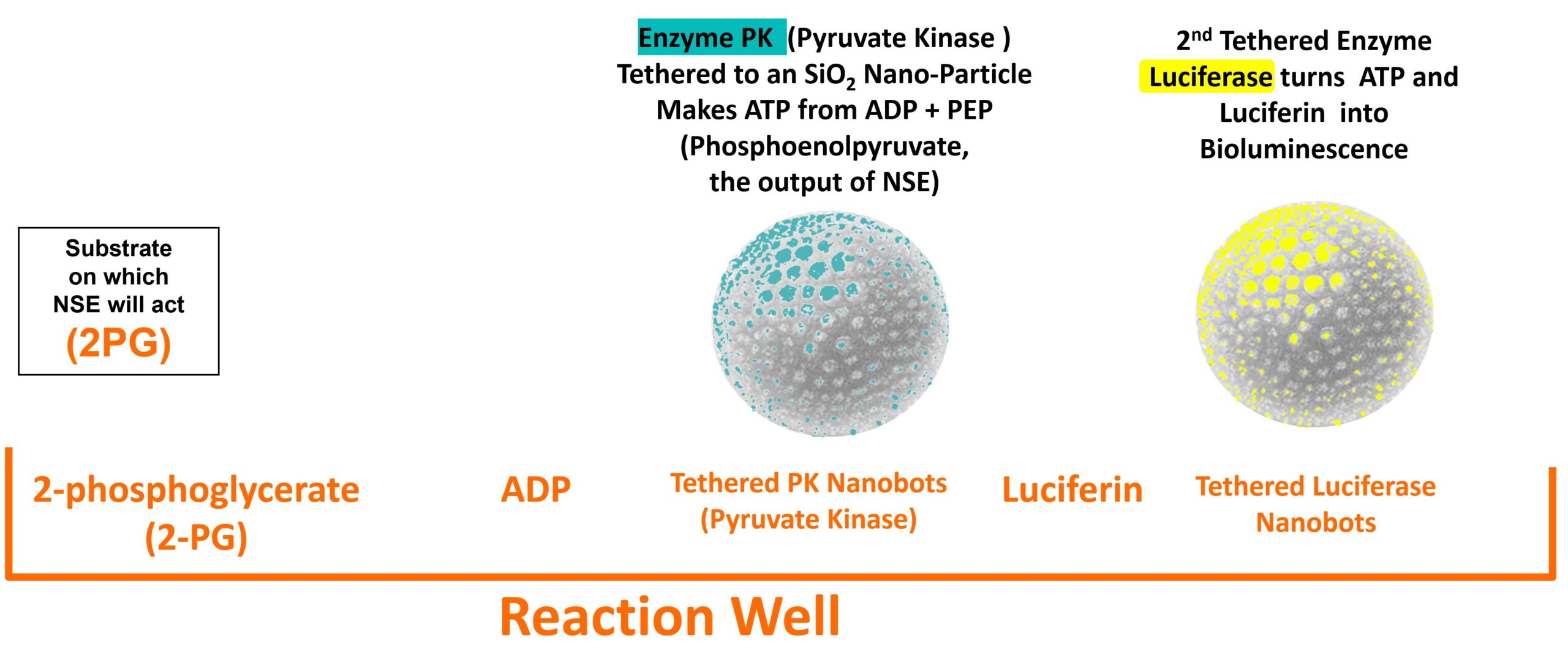
Negative control wells have everything in the reaction well above except for the 2-PG. Positive control wells include a freeze dried specific amount of enolase that will activate when plasma wets the well contents.
The diagram below shows the steps that occur as the catalytic reaction occurs to create luminescence when there is enzymatically active NSE in the plasma sample placed into the reaction well.
- Active NSE molecules that are in the plasma pipetted into the reaction well hydrate the freeze dried contents and begin converting 2PG to PEP at up to a thousand conversions per second per NSE molecule.
- Once the PEP is available the PK nanobots will begin converting the PEP plus ADP to ATP.
- Once the ATP is available, the luciferase nanobots will begin taking the luciferin and ATP in to emit one photon per ATP molecule
If each enzyme can do up to a thousand conversions per second than a nanobot with a thousand enzymes can do a million per second and with a thousand nanobots you get a billion per second. So with the 2PG, ADP and Luciferen saturated in the wells, the limitation on light output is only the amount of active NSE.
Product In Action
The VIDEO (to the right) shows the speed at which this assay works with first results within 30 seconds of insertion in the plate reader.
Current State Of Testing
The NSE-FAST has been tested in animals (see publications) and in two tests in humans. These include the Guthrie Pilot Study in patients presenting with stroke symptoms and a test in Mixed Martial Arts (MMA) fighters.
Descriptions of these pilots and the results obtained can be seen in the below links.
*article in publication.

